Academician Wender from Stanford, Co-founder of N1 Life, Unveils Latest RNA Delivery Vector for Tunable Liver-Targeted Delivery
May 31, 2024
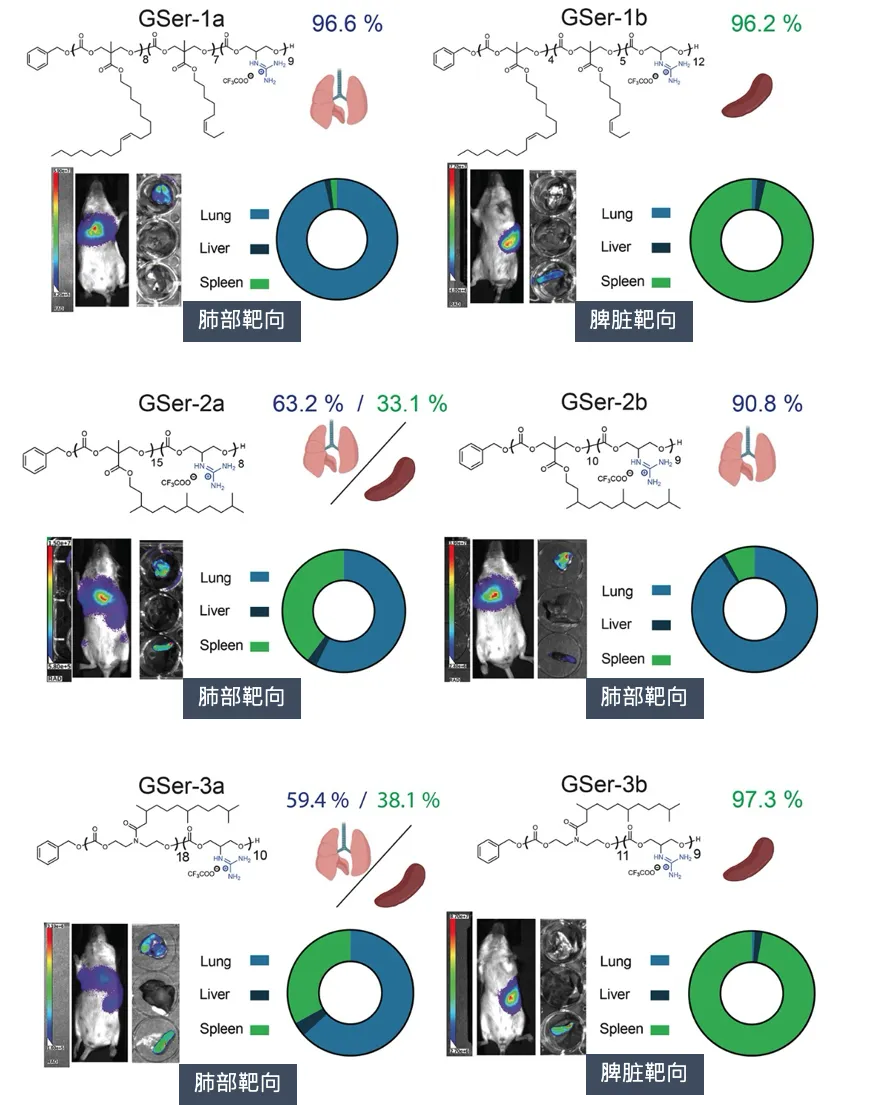
As the corresponding author, Paul A. Wender, co-founder of N1 Life and academician of Stanford University, published the results of cooperation with Professor Robert Waymouth and Professor Howard F. Chang in the well-known academic journal Journal of American Chemistry Society. The title is: "Organ- and Cell-Selective Delivery of mRNA In Vivo Using Guanidinylated Serinol Charge-Altering Releasable Transporters". This article introduces the new progress of Academician Wender's research group in developing organ- and cell-selective mRNA delivery vectors. The new generation of vectors, namely GSer-CARTs vectors, have the characteristics of extrahepatic targeting, controllability, and predictability, and achieve efficient in vivo delivery of circOVA and inhibit tumor growth by inducing immune response.
With the successful development of mRNA vaccine products, the field of RNA drugs has received widespread attention. However, to achieve the widespread application of RNA drugs, one of the major challenges is to develop a safe, effective and scalable delivery system that enables RNA molecules to pass through cell and organ barriers and release RNA at specific sites to exert their effects.
In the past few years, RNA delivery systems represented by lipid nanoparticles (LNPs) have made great progress. Lipid nanoparticles (LNPs) deliver RNA primarily to the liver when injected intravenously and can achieve local delivery when injected intramuscularly, but achieving efficient and selective delivery of RNA to other organs such as the lungs and lymphatic system remains a major technical challenge in the industry.
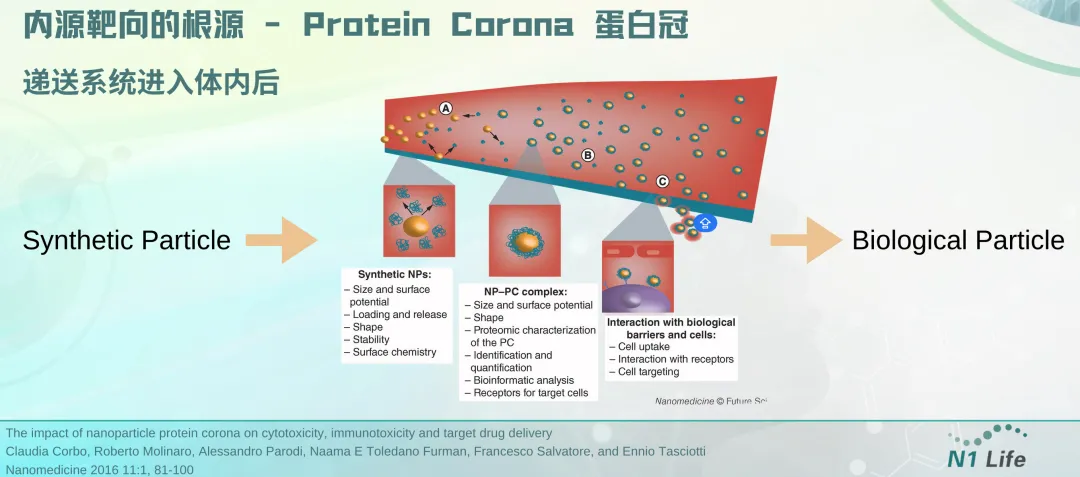
To achieve organ- and cell-selective delivery, one strategy is to modify the LNP surface with receptors that target specific cell surface receptors. This "active targeting" strategy can improve in vivo mRNA delivery to leukocytes, T lymphocytes, and brain cells compared to LNPs without targeting ligands. Targeting ligands that affect biodistribution can also be identified by in vivo library screening. In addition, the "corona effect", which is the modification of the LNP surface upon binding to ligands in the blood, can also effectively participate in organ-selective delivery. Another basic strategy to achieve efficient and selective delivery of mRNA is to design mRNA carriers that lose their cationic charge after in vivo administration. An example of this strategy, Charge-Altering Releasable Transporters (CARTs), was previously reported by the Chang, Levy, Waymouth, and Wender research groups. Wender's laboratory previously reported the early design, subsequent optimization and application of CART vectors. The vectors constructed by step-by-step simple assembly can effectively deliver a variety of RNA molecules including mRNA, siRNA, and siRNA.
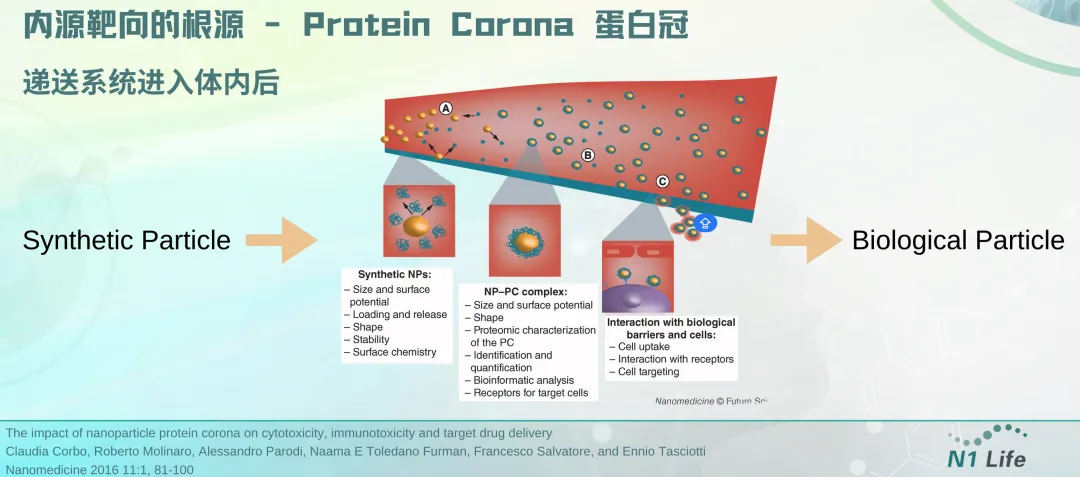
The key role of protein corona: mRNA delivery system undergoes "identity conversion" in vivo to achieve organ targeting | Dr. Janice Zang sharing at BioCon
Characteristics and principles
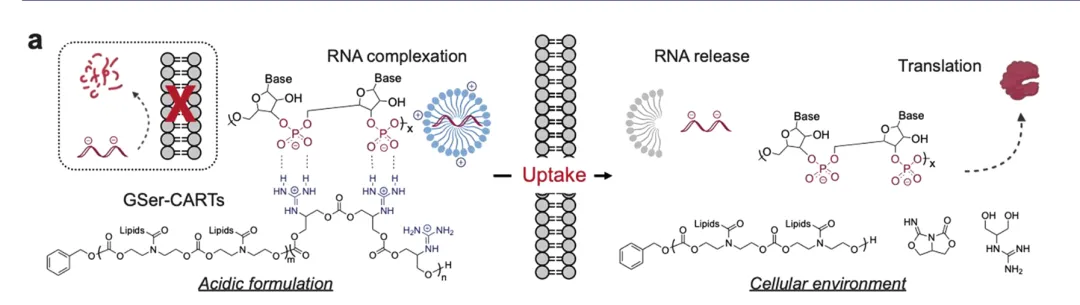
CARTs are block oligomers composed of an initiator (usually a hydroxyl nucleophile, used to initiate polymerization), one or more lipid modules, and a unique cationic α-ammonium ester module, in which the ammonium ester module can electrostatically complex with polyanionic RNA. Under low pH (<5.5), CARTs form a stable complex with mRNA; under physiological pH (~7.4), the cationic ammonium module undergoes oxygen-nitrogen acyl transfer and is irreversibly converted into a non-charged lactam byproduct, thereby triggering the release of polyanionic RNA. CARTs have been used to prepare CAR-NK cells, CAR-T cells, effective SARS-CoV-2 vaccines, and cure cancer and metastatic cancer in mouse models. Unlike LNP systems that are primarily delivered to the liver, CARTs exhibit extrahepatic tropism. For example, CARTs with the addition of fingolimod ligands exhibit selectivity for the spleen; while recently reported CARTs, in the absence of targeting ligands, exhibit significant spleen or lung selectivity. Therefore, CARTs technology provides a new strategy for achieving organ-selective delivery of mRNA. In this article, the Wender research team developed a new guanidine-containing transporter for RNA delivery, which is designed to degrade the guanidine group into neutral byproducts, thereby achieving efficient mRNA release and translation. Based on previous studies of CART carrier molecules, the authors replaced the commonly used charged ammonium cation group with guanidinyl serinol, so it is called GSer-CART.
Figure [1] Biophysical properties and principles of GSer-CART and GSer-CART/mRNA complexes
Research process
First, the research team revealed a new mechanism through the study of model compounds: at low pH (<5.5), the guanidine group forms a double hydrogen bond ring with the carbonate group, and the transporter and mRNA form a stable packaged complex. At high pH, that is, at physiological neutral pH (~7.4), the guanidine group loses its positive charge, thereby eliminating its electrostatic binding with the phosphate group. The guanidine group in the complex can be quickly converted into an electrically neutral byproduct through a cyclization cascade reaction, allowing the two to separate and achieve the release of mRNA in cells.